
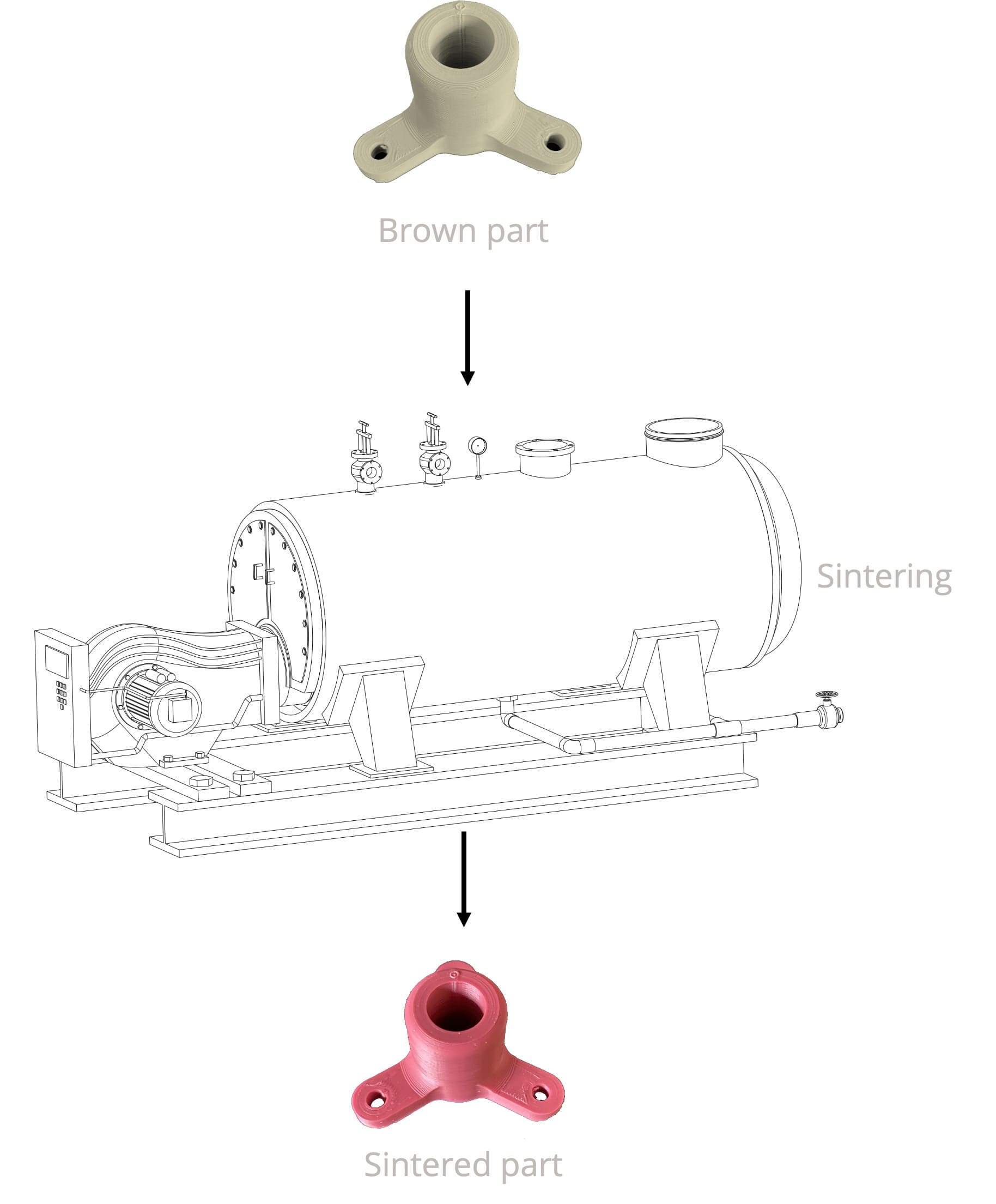
Sintering process
Sintering can be defined as the set of transformations which lead, by heat treatment and without total melting of the material, from an assembly of disjoint grains (the raw part) to a consolidated part (the sintered part).
The welds which occurred between the grains may or may not be accompanied by densification and / or growth of the grains.
We can thus have:
- consolidation without densification (production of ceramic filters) - densification alone (sought for obtaining very fine microstructures);
- densification associated with growth (the most frequent situation).
If no liquid phase appears, sintering is said to be “in solid phase” with two cases:
- non-reactive sintering: a chemical constituent at the start, a part composed of the same constituent at the end,
- reactive sintering: densification is accompanied by one or more chemical reactions between the constituents.
If a liquid phase appears, the sintering is said to be “in the liquid phase”: the liquid phase (minority to maintain the mechanical strength of the part) can come from the simple fusion of a second constituent present (addition of sintering or impurity) or of an eutectic reaction between different constituents.
Depending on whether an external mechanical stress is applied or not, a distinction is made between natural sintering and sintering under load.
Discover Metal and Ceramic 3D printed parts





 Français
Français




